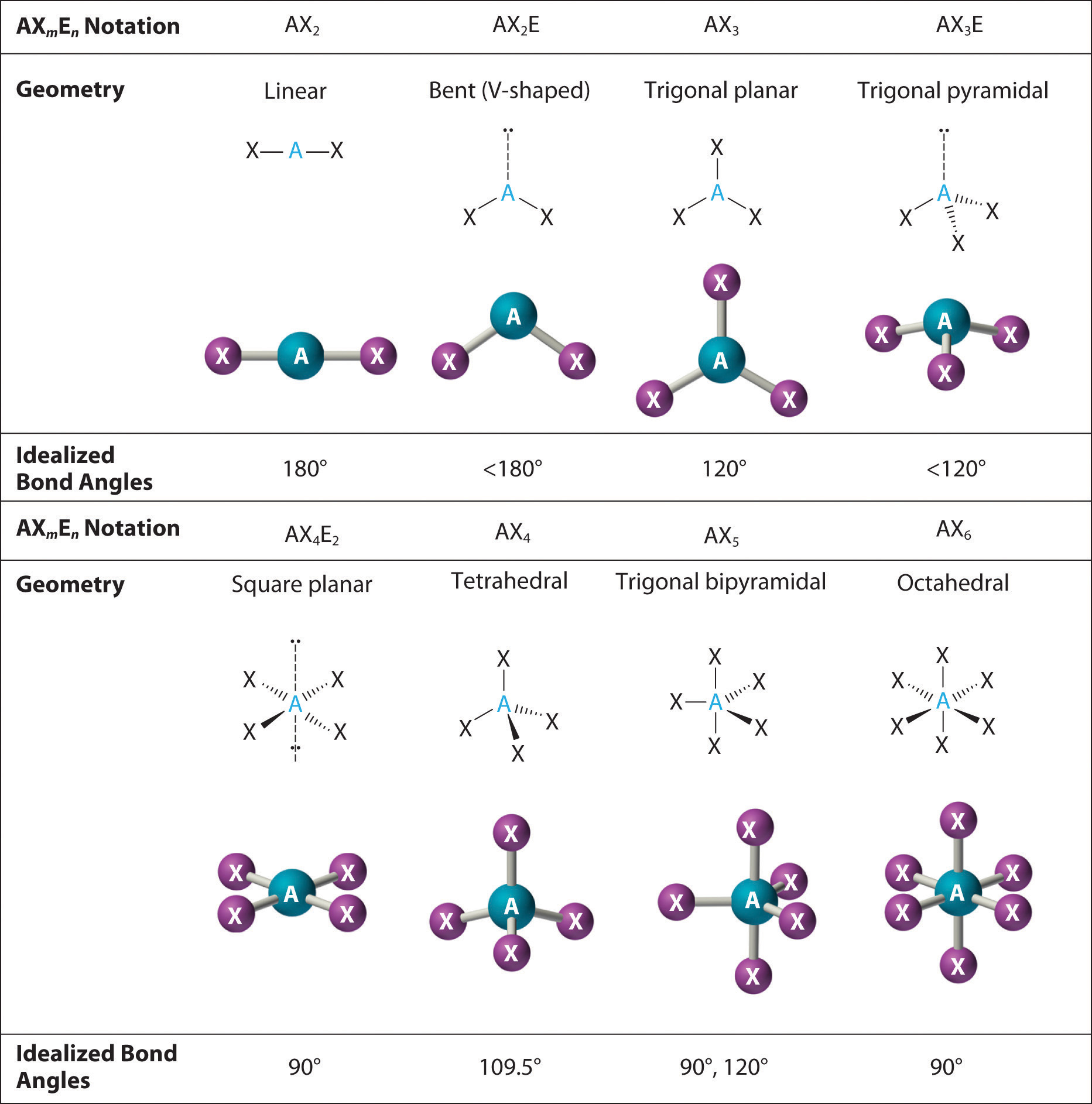
Also 15 lone electron pairs out of 30 remaining electrons get shared equally within 5 fluorine atoms means per fluorine atom has 3 lone electron pairs. 
So, 10 electrons are engaged in forming five bonding pairs with 5 F atoms from total 40 valence electrons of SbF5 molecule. All the valence electrons left will go to fluorine atom till it have electrons to complete the octet. 10 electrons in outer shell instead of 8 electrons. Antimony has an extended octet in SbF5, as has 5 Sb-F bonds i.e. There should be eight electrons on fluorine atom present in outer shell of SbF5 lewis structure. Hence, total electron pairs = 40 ÷ 2 = 20 SbF5 lewis structure octet ruleĪdvertisements Octet rule means having eight electrons in outermost valence shell orbital. There are 40 valence electrons total and it gets divided by the number 2 to get total electron pair count.įormula is: Total electron pairs = total no. Now we have to find the total electron pairs. SbF5 lewis structure forming single bonds between antimony and fluorine So, here thirty valence electrons left for bonding. Therefore, 10 valence electrons were used in drawing SbF5 lewis structure from the total 40 valence electrons present. Here, five single bonds get formed in SbF5 molecule. Now, do the calculation of valence electrons in SbF5 lewis structure, as there are two electrons in 1 single bond. That means five single bonds should be drawn connecting to central antimony atom with five fluorine atoms. So, there is a need to draw a single bond (Sb-F bond) within 1 Sb and 5 F elements to attach them with each other. Next point is the connection of all the atoms by bonding with each other. SbF5 lewis structure showing antimony (Sb) at central position 
That’s why central position is occupied by antimony element covered by five fluorine atom in SbF5 lewis structure. So it seems that electronegative of antimony is lesser than fluorine. In SbF5 compound antimony electronegativity is 2.05 and fluorine’s electronegativity is 3.98. Least electronegative atom from SbF5 structure should be at central position in the structure because they easily share electrons with other atoms attached with it. 
SbF5 lewis structure showing valence electrons So, total valence electrons for SbF5 lewis structure = 5 (Sb) + 7×5 (F5) = 40 So there are five on antimony and seven on fluorine valence electrons respectively. To count the valence electron just check the positions of group F and Sb atoms in periodic table as antimony belongs to 15 th group and fluorine belongs to 17 th group of periodic table. There are 10 bonding electrons and 30 non-bonding electrons in SbF5 molecule.Īdvertisements When we draw SbF5 lewis structure, first thing is to note all the valence electrons present on it. It has 1 antimony and 5 fluorine atoms in the SbF5 lewis structure. SbF5 lewis structure contains 5 fluorine and 1antimony atom in its structure. Molecular geometry of SbF5 is trigonal bipyramidal shape. Molecular weight of SbF5 is 216.752 g mol -1. How to draw lewis structure for SbF5?Ĭhemical formula for antimony pentafluoride is SbF5. SbF5 considered as very dangerous compound for our tissues, because as it burns it can cause gangrene to any person. SbF5 is corrosive on various metals and body tissues. Human eyes and mucous membranes get irritated from its fumes. SbF5 lewis structure contains one antimony (Sb) and five flourine (F) atoms in its structure.Īntimony pentafluoride is an oily liquid form compound, viscous in nature and seems colourless.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
